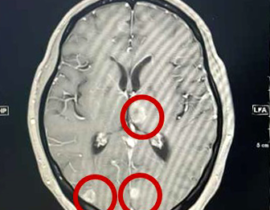
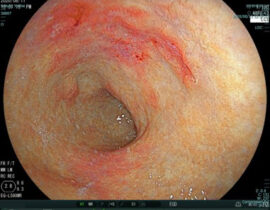
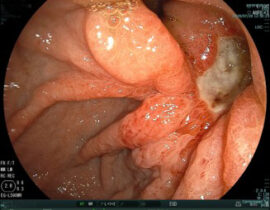
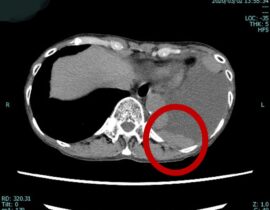
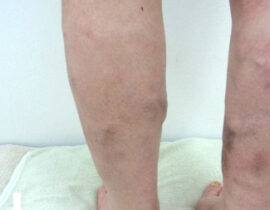
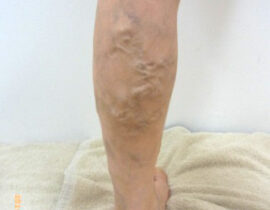
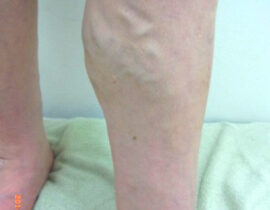
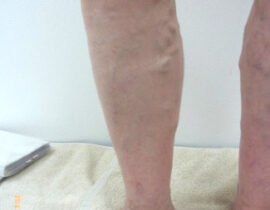
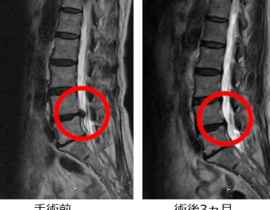
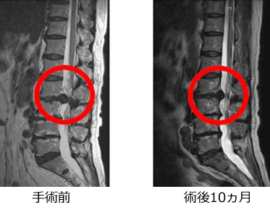
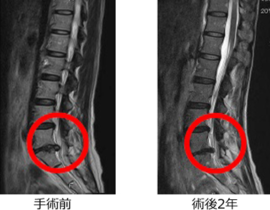
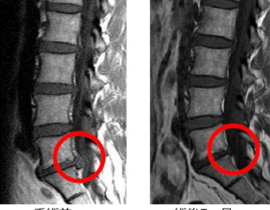
安全性
がんの遺伝子治療「CDC6shRNA治療」
安全性
当たり前のことですが、効かない薬は開発する意味がありません。
しかし、効く薬であっても、重篤な副作用があるものは患者さんに投与できません。新薬を実用化する過程では、有効性と安全性が両立されなければならないのです。
「CDC6 RNAi 治療」が、がん細胞の分裂を停止させたり、がん細胞を自殺させたりする効力を有していたとしても、重篤な副作用や危険性があるならば、現実的にはこの治療を普及させることはできません。
北青山D.CLINICの阿保院長がCDC6 RNAi 治療に出会った時には、極めて新しい治療であったにもかかわらず、安全性を確認するために必要な「試験管での試験」、「小動物から大動物に対する治験」、「健常人に対する治験」、「がん患者さんに対する治験」などを経ないまま、実際に数十人の方がすでに治療を受けている状況でした。CDC6 RNAi 治療で用いる試薬の作成者であるLuo Feng博士からは、基礎実験についての論文は提示されており、試験管での実験や動物実験を行ったとの報告がありましたが、正規の創薬にみられる検証は確認できませんでした。
しかし、
- すでに数十人もの日本人のがん患者さんが中国の青島医科大学(国際的にも最も早く遺伝子治療を手掛けている)で本治療を受け、効果が確認されている。
- 副作用が発生したとしても、一時的な発熱や倦怠感など軽微なものである。
- 全員が進行がんや末期がんであるにもかかわらず、本治療により死期が早まったと考えられる方が皆無である。
といった点から、 阿保院長は臨床医の立場から「安全性と有効性が期待できる治療である」と判断し、Luo Feng博士から提示されたさまざまな論文根拠なども検討した結果、2009年から本治療を本格的に開始しました。
治療の安全性に関する論文
- Polyethylenimine-mediated gene delivery to the lug and therapeutic applications, Sante Di Gioia, Massimo Conese, Drug Design, Development and Therapy, 163-188, 2008/Feb
- Development of purification methods for tumor targeted polyplexes applied in vivo, Julia Fahrmeir, N/A, 1-98,2007
- The promises and pitfalls of RNA interference-based therapeutics, Daniela Castanotto , John J. Rossi, Nature, 2009/Jan/22
- Orally delivered siRNA targeting macrophage Map4k4 suppresses systemic inflammation, Myriam Aouadi, Gregory J. Tesz, Sarah M. Nicoloro, Mengxi Wang, My Chouinard, Ernesto Soto, Gary R. Ostroff, Michael P. Czech, Nature, 1180-1184, 2009/Apr/30
- Evidence of RNAi in humans from systemically administered siRNA via targeted nanoparticles, Mark E. Davis Jonathan E. Zuckerman Chung Hang J. Choi , Davis Seligson, Anthony Tolcher, Christopher A. Alabi, Yun Yen, Jeremy D. Heidel , Antoni Ribas, Nature, 1067-1070, 2010/Apr/15
- Transfusion independence and HMGA2 activation after gene therapy of human β-thalassaemia, Marina Cavazzana-Calvo, Emmanuel Payen, Olivier Negre, Gary Wang, Kathleen Hehir, Floriane Fusil, Julian Down , Maria Denaro, Troy Brady, Karen Westerman, Resy Cavallesco, Beatrix Gillet-Legrand, Laure Caccavelli, Riccardo Sgarra, Nature, 318-322, 2010/Sep/16
- Hematopoietic stem cell gene transfer in a tumor-prone mouse model uncovers low genotoxicity of lentiviral vector integration, Eugenio Montini, Daniela Cesana, Manfred Schmidt, Francesca Sanvito, Maurilio Ponzoni, Cynthia Bartholomae, Lucia Sergi Sergi, Fabrizio Benedicenti, Alessandro Ambrosi, Clelia Di Serio, Claudio Doglioni, Christof von Kalle, Luigi Naldini, Nature Biotechnology, 687-696, 2006/Jun
動物実験レポート
Suppress Proliferation of Implanted Human Breast Bcap-37 Tumors in Nude Mice by AJS2010
WANG, Dechang; LIU, Fusheng; LIU, Chaoyang; ZHANG, Ziqiang
Cancer Institute Chinese Academy of Medical Sciences Department of Pharmacology
Beijing, China
Animal test requested by: LI, Zhong Misa Corporation & Lapian’s Corporation Tokyo, Japan FENG, Luo RNTein Biotech Lab., San Gabriel, California 91776 USA August 30, 2011
Suppress Proliferation of Implanted Human Breast Bcap-37 Tumors in Nude Mice by AJS2010
Abstract Aim: to determine tumor suppressing effects by direct intra-tumor injection of AJS2010, the novel recombinant lentiviral vectors producing human CDC6 short haipin RNA (shRNA), human p16INK4a, and human PTEN.
Methods: Seventy female nude mice were implanted with human breast Bcap-37 tumors and kept for breeding for 6 days. The nude mice were formed into 7 groups: Group 1: blank control (injecting 1xPBS, 50 μl per animal); Group 2: recombinant interferon γ positive control (injecting 25x10k units per animal); Group 3, 4, 5: injecting AJS2010 at 27.5 μg, 88 μg, and 275 μg of proteins per animal, respectively; Group 6: blank control of animals kept for breeding for extended period (injecting 1xPBS, 50 μl per animal); and Group 7: injecting AJS2010 at 275 μg of proteins per animal kept for breeding for extended period. Intra-tumor injection of 50 μl in volumes was performed for each animal with the injected liquids containing specific amounts of proteins in AJS2010, and the injections were done twice a week. Animals in Group 1-5 were put for euthanasia by cervical dislocation 48 hours after final dosage, while animals in Group 6 and 7 were kept to examine tumor growth until some dead mouse was evident. The animals in Group 6 and 7 were then euthanasia to kill. The body weight was determined prior AJS2010 injection and before the euthanasia to kill the animal. The tumors were removed from the dead mice and fixed and processed for pathological examination.
Results: There were no abnormal behaviors during six injections in all seven groups of animals, and there was no single animal death due to the toxic effect of AJS2010. The body weight in Group 1-5 was gained for two grams, while in Group 6 and 7 one gram. The weight of tumor mass in group with injection of AJS2010 at 275 μg of proteins per animal was significantly less than that of negative control (p<0.02), the efficiency of inhibition of tumor proliferation was about 34.7%. The size of the tumor in group with injection of AJS201 at 275 μg of proteins per animal was significantly smaller than those in negative control (p<0.02). Tissue sections from the tumors showed fibrosis developed significantly in group of recombinant interferon γ and in groups of 3-5 in comparison of negative control (Group 1) (p<0.02 or p<0.001).
Discussion: Results from intra-tumor injection of AJS2010 at 88 μg of or 275 μg of proteins per animal show stronger inhibition of tumor proliferation in comparison of that of recombinant interferon γ. The decreased tumor size along with the stronger reactions in developing fibrosis in tumor tissues is evident. We consider that the efficacy of AJS2010 in inhibiting implanted human breast Bcap-37 tumors is related to the development of fibrosis in tumor tissues. Moreover, there is no immune reaction observed during repeatedly injecting AJS2010 in testing animals.
Materials and Methods
1 Reagents:
(A) AJS2010: from RNTein Biotech Lab. (San Gabriel, California, USA), CPP-T11026 (0001-0009), 1 ml/vial. Protein concentration: 5.5 mg/ml. Stored at -80°C.
(B) Interferon γ: recombinant, 100x10k units/vial, Shanghai Kai-Mao Bioproducts, Ltd., Shanghai, China. Government Permission number: 10980084, Lot number: 20100909. All of mice were kept for breeding in animal room with an exhaust-air ventilation system. The room was with light on for 12 hours every day and the temperature was kept at 18-22°C with relative humidity around 40-60%. Animals were fed with Clean grade foods with production permission number Beijing-Veterinary 2000-15. The drink water was from deionized ultrafiltered ddH2O. Food and drinking water were given freshly everyday, and the animal room was cleaned by specific housekeeping personal. All of mice were checked with no any abnormality observed before being treated in the animal tests.
3 Human tumor strain: strain Bcap-37 is derived from human breast carcinoma, and cultured and kept in this animal facility.
4 Tumor implantation: Working in a Biosafty cabinet under sterilization condition, the tumor was taken out of host animal, and the tumor tissues were rinsed with 1xPBS. Those tumor areas with fine growth without decay were collected, and small pieces of blocks in 3 mm3 were made with a scalpel. The tumor blocks were rinsed with 1xPBS, and implanted one by one underneath the skin of each mouse’s right armpit with a tubing needle. Some anti-germ reagent was administered to the cut at skin before the cut being covered with sterilized cotton bandage.
5 Grouping and injecting: The growing up tumors could be touched 6 days after implantation. The animals were formed into seven groups, 10 mice in each group:
- Blank negative control: 1xPBS, 50 μl per animal, twice a week;
- Positive control, recombinant interferon γ, 25x10k units per animal in 50 volumes, twice a week;
- AJS2010 injection at 27.5 μg of proteins per animal in 50 volumes, twice a week;
- AJS2010 injection at 88.0 μg of proteins per animal in 50 volumes, twice a week;
- AJS2010 injection at 275 μg of proteins per animal in 50 volumes, twice a week;
- Blank negative control: 1xPBS, 50 μl per animal, twice a week, for extended period of treatment;
- AJS2010 injection at 275 μg of proteins per animal in 50 volumes, twice a week, for extended period of treatment.
Five mice in each group were kept in one cage and total of 14 cages were used during the experiments. The use of AJS2010 was done according to the manual provided by the RNTein Biotech lab. (San Gabriel, California, USA). Each injection volume was brought to 50 μl using 1xPBS though there were different concentrations of AJS1010 proteins in the injection. The 100-microleter syringes were used for the intra-tumor injection. Multiple spots inside tumor foci were injected under sterilization condition. About 5-10 μl of injection were at each spot. Injection was done twice a week for consecutive three weeks. The concentrations of proteins in AJS2010 used in the injections were recommended by the RNTein Biotech Lab. (San Gabriel, California, USA). The values with different concentration ratio were 0.31-0.32 among Group 3-5 and Group 7.
(6) The measurement of size of tumors: The measurement of length and width of each tumor was done twice a week after the injection. A standard caliper was used to take the measurement. Tumors grown in Group 6 and 7 animals were also measured in their sizes twice a week after the treatment.
(7) Euthanasia to put the testing animal death: Animals in Group 1-5 are in euthanasia to kill by cervical dislocation 48 hours after the final dosage. The time for euthanasia to kill all of the animals in Group 6 and 7 was not started until the death of some mouse was evident. The tumors from all of the dead mice were collected and processed for pathological analysis.
(8) Calculation of testing mouse’s body weight: The mouse’s body weight was measured at the time of the AJS2010 injection (tumor carrying body weight). Body weight for the dead mouse was measured after the tumor was removed from the body (net body weight). Net body weight=Tumor carrying body weight – Tumor mass weight.
(9) Efficacy:
(a) Efficiency of the inhibition of tumor proliferation was calculated as:
The Efficiency of tumor proliferation inhibition (I) = [1- (T)/ (C)] x100%
T: Average tumor mass weight in Group 3-5, or 7
C: Average tumor mass weight in Group 1, or 6
The treatment was validated as “EFFECTIVE” when “I” was high enough and the p < 0.05 is evident.
(B) Calculation of tumor size: Tumor size = (Tumor length x Tumor width)2/2.
(C) Tissue section and evaluation: The marker “-“, for negative; “+”, for low grade of fibrosis; “++” for modest fibrosis”, and “+++” for highest grade of fibrosis were used in the evaluation of fibrosis of tumor tissues. The number 0, 1, 2, and 3 corresponding to the markers respectively were integrated for the computation and statistics analysis.
Results
All of the available AJS2010 had been used in total six injections in three weeks. There were no acute toxic effect on the testing animals during the injection, nor was any animal died due to the injection of AJS2010. Moreover, the testing animals gained body weights during the treatment. The average body weight (in gram), average weight of tumors (in gram), average size of tumors (mm3) measured in all of the animals in each group are listed in Table 1 and in Figure 1 to 5. Fibrosis of tumor tissues is listed in Table 2. By the end of animal experiments, the animals gain body weight average 2 grams in groups 1 to 5, average 1 gram in groups 6 and 7. The inhibition of implanted tumor proliferation is 9,80% in Group 3, 24.84% in Group 4, and 34.70% in Group 5. When 275 μg of proteins in AJS2010 are used per animal, the average weight of tumors is significantly less than that of the negative control (p<0.02). Compared among the Group 3, 4 and 5, the inhibition is significant when 275 μg of proteins in AJS2010 are used per animal (p<0.01). The results are similar to that of recombinant interferon γ (Group 2). The average weight of tumors in Group 7 is significantly less than that of negative control in Group 6 (p<0.05). It is significant in developing fibrosis of tumor tissues in Group 3 to 5 and in Group 7 in comparison of negative control in Group 1 and 6. The section of tumor tissues reveals elevated levels of fibrosis developed in tumor tissues by injecting AJS2010 (p<0.01 or p<0.001).
Conclusion
There is no acute toxic effect on all of testing mice by intra-tumor injection of AJS2010. In addition, there is no animal death during the AJS2010 treatment. The death of one mouse in the Group 6 for extended period of treatment could be the results of progression of implanted tumor. The concentrations of proteins in AJS2010 used in the injections reveal efficacy of the reagent in inhibiting tumor proliferation with reduction of tumor size and weight of implanted tumors. Relatively better effective than positive control recombinant interferon γ on inhibiting tumor proliferation has been evident. Higher concentrations of proteins in AJS2010 used in the injections results in strong fibrosis developed in tumor tissues, which suppresses the tumor growth. The fact that by repeatedly injecting AJS2010, the tumor growth is continuously slow down during three week treatment indicates that there is no immune reactions against the reagent. If combined with some kinds of chemotherapies, the application of non-cytotoxic AJS2001 could produce much better effect in treatment of cancer patients. It is necessary to do such investigation in clinical applications. Moreover, how high the concentration of proteins in AJS2010 can be used without obvious side effect is obscure. More animal testing regarding this matter should be done in the future. We would thank LAPIANs Corporation in Tokyo, Japan for supporting these animal experiments. Enclosure: Tables and Figures. Table I The effect of AJS2010 intra-tumor injection on implanted tumor growth (Compare average body weight of mice, average weight of tumor, and average size of tumor). Table II Fibrosis of tumor tissue after the injection of AJS2010. Figure I The effect of AJS2010 on tumor growth (Tumor growth curve). Figure II Inhibition of tumor proliferation by AJS2010 (Compare average weight of the tumor). Figure III Inhibition of tumor growth by AJS2010 (Compare average tumor size). Figure IV Extensive observation of effect of AJS2010 on tumor growth (Tumor growth curve in 50 days). Figure V Extensive observation of effect of AJS2010 on tumor growth (Average weight of the tumor in extensive treatment).